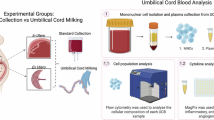
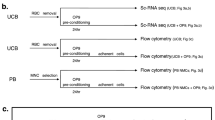
Abstract
Neonatal hypoxic-ischemic encephalopathy (HIE) in preterm infants lacks effective treatment and remains a leading cause of long-term neurological impairment. Autologous umbilical cord blood (UCB)-derived CD34⁺ cell therapy shows therapeutic potential; however, limited UCB volume in preterm infants often yields insufficient cells for clinical use. Recent advances in ex vivo expansion using chemically defined, albumin- and cytokine-free systems enable efficient proliferation of CD34⁺ hematopoietic stem cells. We evaluated the safety and efficacy of intravenously administered, ex vivo expanded CD34⁺ cells derived from human UCB in a mouse model of preterm HIE. Human UCB-derived CD34⁺ cells were cultured for 10 days using a chemically defined, serum- and cytokine-free expansion protocol. Mice with unilateral hypoxic-ischemic injury on postnatal day 5 received expanded or unexpanded CD34⁺ cells (1 × 10⁵ cells per animal) 48 h after injury. Expanded CD34⁺ cells were well tolerated and improved sensorimotor asymmetry, as assessed by the cylinder test, and reduced astrocyte and microglial activation in the injured brain compared with untreated HIE mice. Therapeutic effects did not significantly differ between expanded and unexpanded CD34⁺ cells. Ex vivo expansion under chemically defined conditions may enable autologous CD34⁺ cell therapy for preterm infants with limited UCB volume and represents a promising approach for regenerative treatment of neonatal brain injury.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. Due to confidentiality agreements, some information related to the cell materials provided by Celaid Therapeutics is not publicly available.
References
Mo, Y. et al. Early-stage effect of HIBD on neuro-motor function and organic composition of neurovascular units in neonatal rats. Front. Neurosci. 17, 1242936. https://doi.org/10.3389/fnins.2023.1242936 (2023).
Li, J., McDonald, C. A., Fahey, M. C., Jenkin, G. & Miller, S. L. Could cord blood cell therapy reduce preterm brain injury? Front. Neurol. 5, 200. https://doi.org/10.3389/fneur.2014.00200 (2014).
Bennet, L. et al. Cell therapy for neonatal hypoxia-ischemia and cerebral palsy. Ann. Neurol. 71, 589–600. https://doi.org/10.1002/ana.22670 (2012).
Gunn, A. J. & Bennet, L. Brain cooling for preterm infants. Clin. Perinatol. 35, 735–748. https://doi.org/10.1016/j.clp.2008.07.012 (2008).
de Almeida, M. F. et al. Hypothermia and early neonatal mortality in preterm infants. J. Pediatr. 164, 271–275e271. https://doi.org/10.1016/j.jpeds.2013.09.049 (2014).
Hall, A. A. et al. Human umbilical cord blood cells directly suppress ischemic oligodendrocyte cell death. J. Neurosci. Res. 87, 333–341. https://doi.org/10.1002/jnr.21857 (2009).
Zhu, L. H. et al. Improvement of human umbilical cord mesenchymal stem cell transplantation on glial cell and behavioral function in a neonatal model of periventricular white matter damage. Brain Res. 1563, 13–21. https://doi.org/10.1016/j.brainres.2014.03.030 (2014).
Ahn, S. Y. et al. Mesenchymal stem cells prevent hydrocephalus after severe intraventricular hemorrhage. Stroke 44, 497–504. https://doi.org/10.1161/STROKEAHA.112.679092 (2013).
Yawno, T. et al. Human amnion epithelial cells reduce fetal brain injury in response to intrauterine inflammation. Dev. Neurosci. 35, 272–282. https://doi.org/10.1159/000346683 (2013).
Vinukonda, G. & La Gamma, E. F. Emerging therapies for brain recovery after IVH in neonates: cord blood derived mesenchymal stem cells (MSC) and unrestricted somatic stem cells (USSC). Semin Perinatol. 46, 151598. https://doi.org/10.1016/j.semperi.2022.151598 (2022).
Cotten, C. M. et al. Feasibility of autologous cord blood cells for infants with hypoxic-ischemic encephalopathy. J. Pediatr. 164, 973–979. https://doi.org/10.1016/j.jpeds.2013.11.036 (2014). .e971.
Tsuji, M. et al. Autologous cord blood cell therapy for neonatal hypoxic-ischaemic encephalopathy: a pilot study for feasibility and safety. Sci. Rep. 10, 4603. https://doi.org/10.1038/s41598-020-61311-9 (2020).
Rocha, V. et al. Association of drug metabolism gene polymorphisms with toxicities, graft-versus-host disease and survival after HLA-identical sibling hematopoietic stem cell transplantation for patients with leukemia. Leukemia 23, 545–556. https://doi.org/10.1038/leu.2008.323 (2009).
Majka, M. et al. Numerous growth factors, cytokines, and chemokines are secreted by human CD34(+) cells, myeloblasts, erythroblasts, and megakaryoblasts and regulate normal hematopoiesis in an autocrine/paracrine manner. Blood 97, 3075–3085. https://doi.org/10.1182/blood.v97.10.3075 (2001).
Shizuru, J. A., Negrin, R. S. & Weissman, I. L. Hematopoietic stem and progenitor cells: clinical and preclinical regeneration of the hematolymphoid system. Annu. Rev. Med. 56, 509–538. https://doi.org/10.1146/annurev.med.54.101601.152334 (2005).
Baraniak, P. R. & McDevitt, T. C. Stem cell paracrine actions and tissue regeneration. Regen Med. 5, 121–143. https://doi.org/10.2217/rme.09.74 (2010).
Czechowicz, A. & Weissman, I. L. Purified hematopoietic stem cell transplantation: the next generation of blood and immune replacement. Hematol. Oncol. Clin. North. Am. 25, 75–87. https://doi.org/10.1016/j.hoc.2010.11.006 (2011).
Aguila, H. L. et al. From stem cells to lymphocytes: biology and transplantation. Immunol. Rev. 157, 13–40. https://doi.org/10.1111/j.1600-065X.1997.tb00971.x (1997).
Ohshima, M. et al. Evaluations of intravenous administration of CD34 + human umbilical cord blood cells in a mouse model of neonatal hypoxic-ischemic encephalopathy. Dev. Neurosci. 38, 331–341. https://doi.org/10.1159/000454830 (2016).
Yu, Y. et al. Effects of human umbilical cord blood CD34(+) cell transplantation in neonatal hypoxic-ischemia rat model. Brain Dev. 41, 173–181. https://doi.org/10.1016/j.braindev.2018.08.007 (2019).
Paton, M. C. B. et al. Updates on neonatal cell and novel therapeutics: proceedings of the Second Neonatal Cell Therapies Symposium (2024) Pediatr. Res. https://doi.org/10.1038/s41390-025-03856-x (2025).
Boitano, A. E. et al. Aryl hydrocarbon receptor antagonists promote the expansion of human hematopoietic stem cells. Science 329, 1345–1348. https://doi.org/10.1126/science.1191536 (2010).
Fares, I. et al. Pyrimidoindole derivatives are agonists of human hematopoietic stem cell self-renewal. Science 345, 1509–1512. https://doi.org/10.1126/science.1256337 (2014).
Cohen, S. et al. Hematopoietic stem cell transplantation using single UM171-expanded cord blood: a single-arm, phase 1–2 safety and feasibility study. Lancet Haematol. 7, e134–e145. https://doi.org/10.1016/S2352-3026(19)30202-9 (2020).
Wagner, J. E. et al. Phase I/II trial of StemRegenin-1 expanded umbilical cord blood hematopoietic stem cells supports testing as a stand-alone graft. Cell. Stem Cell. 18, 144–155. https://doi.org/10.1016/j.stem.2015.10.004 (2016).
Wilkinson, A. C. et al. Long-term ex vivo haematopoietic-stem-cell expansion allows nonconditioned transplantation. Nature 571, 117–121. https://doi.org/10.1038/s41586-019-1244-x (2019).
Sakurai, M. et al. Chemically defined cytokine-free expansion of human haematopoietic stem cells. Nature 615, 127–133. https://doi.org/10.1038/s41586-023-05739-9 (2023).
Penny, T. R., Jenkin, G., Miller, S. L. & McDonald, C. A. Umbilical cord blood derived cell expansion: a potential neuroprotective therapy. Stem Cell. Res. Ther. 15, 234. https://doi.org/10.1186/s13287-024-03830-0 (2024).
Saiyin, T. et al. Clinical outcomes of umbilical cord blood transplantation using ex vivo expansion: a systematic review and meta-analysis of controlled studies. Transpl. Cell. Ther. 29, 129e121–129e129. https://doi.org/10.1016/j.jtct.2022.11.007 (2023).
Chua, S. J. et al. The effect of umbilical cord blood cells on outcomes after experimental traumatic spinal cord injury. Spine (Phila. PA) 35, 1520–1526. (1976). https://doi.org/10.1097/BRS.0b013e3181c3e963 (2010).
Whiteley, J. et al. An expanded population of CD34 + cells from frozen banked umbilical cord blood demonstrate tissue repair mechanisms of mesenchymal stromal cells and circulating angiogenic cells in an ischemic hind limb model. Stem Cell. Rev. Rep. 10, 338–350. https://doi.org/10.1007/s12015-014-9496-1 (2014).
Tsuji, M. et al. Effects of intravenous administration of umbilical cord blood CD34(+) cells in a mouse model of neonatal stroke. Neuroscience 263, 148–158. https://doi.org/10.1016/j.neuroscience.2014.01.018 (2014).
Tanaka, E. et al. Dose-dependent effect of intravenous administration of human umbilical cord-derived mesenchymal stem cells in neonatal stroke mice. Front. Neurol. 9, 133. https://doi.org/10.3389/fneur.2018.00133 (2018).
Yasuhara, T. et al. Mannitol facilitates neurotrophic factor up-regulation and behavioural recovery in neonatal hypoxic-ischaemic rats with human umbilical cord blood grafts. J. Cell. Mol. Med. 14, 914–921. https://doi.org/10.1111/j.1582-4934.2008.00671.x (2010).
Donega, V. et al. Intranasal mesenchymal stem cell treatment for neonatal brain damage: long-term cognitive and sensorimotor improvement. PLoS One 8, e51253. https://doi.org/10.1371/journal.pone.0051253 (2013).
Ben-Hur, T. Immunomodulation by neural stem cells. J. Neurol. Sci. 265, 102–104. https://doi.org/10.1016/j.jns.2007.05.007 (2008).
Li, J. et al. Preterm white matter brain injury is prevented by early administration of umbilical cord blood cells. Exp. Neurol. 283, 179–187. https://doi.org/10.1016/j.expneurol.2016.06.017 (2016).
Purcell, E. et al. Factors influencing the efficacy of umbilical cord blood-derived cell therapy for perinatal brain injury. Stem Cells Transl. Med. 12, 125–139. https://doi.org/10.1093/stcltm/szad006 (2023).
Sato, Y. & Tsuji, M. Diverse actions of cord blood cell therapy for hypoxic-ischemic encephalopathy. Pediatr. Int. 63, 497–503. https://doi.org/10.1111/ped.14604 (2021).
Qi, L. et al. Advances in mesenchymal stem cell-centered stem cell therapy in the treatment of hypoxic-ischemic injury. Int. Immunopharmacol. 143, 113430. https://doi.org/10.1016/j.intimp.2024.113430 (2024).
Serrenho, I. et al. Stem cell therapy for neonatal hypoxic-ischemic encephalopathy: a systematic review of preclinical studies. Int. J. Mol. Sci. 22, 63142. https://doi.org/10.3390/ijms22063142 (2021).
Asgari Taei, A., Khodabakhsh, P., Nasoohi, S., Farahmandfar, M. & Dargahi, L. Paracrine effects of mesenchymal stem cells in ischemic stroke: opportunities and challenges. Mol. Neurobiol. 59, 6281–6306. https://doi.org/10.1007/s12035-022-02967-4 (2022).
Voermans, C. et al. In vitro migratory capacity of CD34 + cells is related to hematopoietic recovery after autologous stem cell transplantation. Blood 97, 799–804. https://doi.org/10.1182/blood.v97.3.799 (2001).
Kong, Y. et al. Increased reactive oxygen species and exhaustion of quiescent CD34-positive bone marrow cells may contribute to poor graft function after allotransplants. Oncotarget 7, 30892–30906. https://doi.org/10.18632/oncotarget.8810 (2016).
Geissler, M., Dinse, H. R., Neuhoff, S., Kreikemeier, K. & Meier, C. Human umbilical cord blood cells restore brain damage induced changes in rat somatosensory cortex. PLoS One. 6, e20194. https://doi.org/10.1371/journal.pone.0020194 (2011).
Rosenkranz, K. et al. Transplantation of human umbilical cord blood cells mediated beneficial effects on apoptosis, angiogenesis and neuronal survival after hypoxic-ischemic brain injury in rats. Cell. Tissue Res. 348, 429–438. https://doi.org/10.1007/s00441-012-1401-0 (2012).
Pimentel-Coelho, P. M. et al. Human cord blood transplantation in a neonatal rat model of hypoxic-ischemic brain damage: functional outcome related to neuroprotection in the striatum. Stem Cells Dev. 19, 351–358. https://doi.org/10.1089/scd.2009.0049 (2010).
Wasielewski, B., Jensen, A., Roth-Härer, A., Dermietzel, R. & Meier, C. Neuroglial activation and Cx43 expression are reduced upon transplantation of human umbilical cord blood cells after perinatal hypoxic-ischemic injury. Brain Res. 1487, 39–53. https://doi.org/10.1016/j.brainres.2012.05.066 (2012).
Rosenkranz, K., Tenbusch, M., May, C., Marcus, K. & Meier, C. Changes in interleukin-1 alpha serum levels after transplantation of umbilical cord blood cells in a model of perinatal hypoxic-ischemic brain damage. Ann. Anat. 195, 122–127. https://doi.org/10.1016/j.aanat.2012.09.003 (2013).
Haynes, R. L. et al. Oxidative and nitrative injury in periventricular leukomalacia: a review. Brain Pathol. 15, 225–233. https://doi.org/10.1111/j.1750-3639.2005.tb00525.x (2005).
Zhou, L. et al. Feasibility and safety of autologous cord blood derived cell administration in extremely preterm infants: a single-centre, open-label, single-arm, phase I trial (CORD-SaFe study). EBioMedicine 111, 105492. (2025). https://doi.org/10.1016/j.ebiom.2024.105492
Schmidt-Lucke, C. et al. Reduced number of circulating endothelial progenitor cells predicts future cardiovascular events: proof of concept for the clinical importance of endogenous vascular repair. Circulation 111, 2981–2987. https://doi.org/10.1161/CIRCULATIONAHA.104.504340 (2005).
Yip, H. K. et al. Level and value of circulating endothelial progenitor cells in patients after acute ischemic stroke. Stroke 39, 69–74. https://doi.org/10.1161/STROKEAHA.107.489401 (2008).
Leonard, A. et al. Bone marrow characterization in sickle cell disease: inflammation and stress erythropoiesis lead to suboptimal CD34 recovery. Br. J. Haematol. 186, 286–299. https://doi.org/10.1111/bjh.15902 (2019).
Zarrabi, M. et al. The safety and efficacy of umbilical cord blood mononuclear cells in individuals with spastic cerebral palsy: a randomized double-blind sham-controlled clinical trial. BMC Neurol. 22 https://doi.org/10.1186/s12883-022-02636-y (2022).
Hagberg, H., Peebles, D. & Mallard, C. Models of white matter injury: comparison of infectious, hypoxic-ischemic, and excitotoxic insults. Ment. Retard. Dev. Disabil. Res. Rev. 8, 30–38. https://doi.org/10.1002/mrdd.10007 (2002).
Rice, J. E., Vannucci, R. C. & Brierley, J. B. The influence of immaturity on hypoxic-ischemic brain damage in the rat. Ann. Neurol. 9, 131–141. https://doi.org/10.1002/ana.410090206 (1981).
Ohshima, M., Tsuji, M., Taguchi, A., Kasahara, Y. & Ikeda, T. Cerebral blood flow during reperfusion predicts later brain damage in a mouse and a rat model of neonatal hypoxic-ischemic encephalopathy. Exp. Neurol. 233, 481–489. https://doi.org/10.1016/j.expneurol.2011.11.025 (2012).
Dunn, A. K., Bolay, H., Moskowitz, M. A. & Boas, D. A. Dynamic imaging of cerebral blood flow using laser speckle. J. Cereb. Blood Flow. Metab. 21, 195–201. https://doi.org/10.1097/00004647-200103000-00002 (2001).
Forrester, K. R., Stewart, C., Tulip, J., Leonard, C. & Bray, R. C. Comparison of laser speckle and laser Doppler perfusion imaging: measurement in human skin and rabbit articular tissue. Med. Biol. Eng. Comput. 40, 687–697. https://doi.org/10.1007/BF02345307 (2002).
Taguchi, A. et al. Administration of CD34 + cells after stroke enhances neurogenesis via angiogenesis in a mouse model. J. Clin. Invest. 114, 330–338. https://doi.org/10.1172/JCI20622 (2004).
Uemura, M., Kasahara, Y., Nagatsuka, K. & Taguchi, A. Cell-based therapy to promote angiogenesis in the brain following ischemic damage. Curr. Vasc Pharmacol. 10, 285–288. https://doi.org/10.2174/157016112799959369 (2012).
Ohshima, M. et al. Intraperitoneal and intravenous deliveries are not comparable in terms of drug efficacy and cell distribution in neonatal mice with hypoxia-ischemia. Brain Dev. 37, 376–386. https://doi.org/10.1016/j.braindev.2014.06.010 (2015).
Tsuji, M., Wilson, M. A., Lange, M. S. & Johnston, M. V. Minocycline worsens hypoxic-ischemic brain injury in a neonatal mouse model. Exp. Neurol. 189, 58–65. https://doi.org/10.1016/j.expneurol.2004.01.011 (2004).
Tsuji, M., Taguchi, A., Ohshima, M., Kasahara, Y. & Ikeda, T. Progesterone and allopregnanolone exacerbate hypoxic-ischemic brain injury in immature rats. Exp. Neurol. 233, 214–220. https://doi.org/10.1016/j.expneurol.2011.10.004 (2012).
Tsuji, M. et al. A novel reproducible model of neonatal stroke in mice: comparison with a hypoxia-ischemia model. Exp. Neurol. 247, 218–225. https://doi.org/10.1016/j.expneurol.2013.04.015 (2013).
Ohshima, M. et al. Mild intrauterine hypoperfusion reproduces neurodevelopmental disorders observed in prematurity. Sci. Rep. 6, 39377. https://doi.org/10.1038/srep39377 (2016).
Ogawa, Y., Tsuji, M., Tanaka, E., Miyazato, M. & Hino, J. Bone morphogenetic protein (BMP)-3b gene depletion causes high mortality in a mouse model of neonatal hypoxic-ischemic encephalopathy. Front. Neurol. 9, 397. https://doi.org/10.3389/fneur.2018.00397 (2018).
van Velthoven, C. T. et al. Mesenchymal stem cells restore cortical rewiring after neonatal ischemia in mice. Ann. Neurol. 71, 785–796. https://doi.org/10.1002/ana.23543 (2012).
Acknowledgements
We gratefully acknowledge the support of Celaid Therapeutics, who provided both the cell materials and funding for this research.
Funding
This study was financially supported by Celaid Therapeutics, which also provided the cells used in the experiments. The funder had no influence on the study design, data collection and analysis, decision to publish, or manuscript preparation. Additional support was provided by JSPS KAKENHI (Grant Number JP222640019).
Author information
Authors and Affiliations
Contributions
Natsumu Arai: Collection and/or assembly of data (behavioral tests, etc.), data analysis and interpretation, figure preparation, manuscript writing. Emi Tanaka: Collection and/or assembly of data (animal model and behavioral tests, etc.), data analysis and interpretation, study design, critical manuscript review. Satoshi Ohnishi: Project supervision, critical revision of the manuscript for important intellectual content. Takashi Hamazaki: Project supervision, critical revision of the manuscript for important intellectual content. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
This study was financially supported by Celaid Therapeutics, which also provided the cells used in the experiments. The funder had no influence on the study design, data collection and analysis, decision to publish, or manuscript preparation. Additional support was provided by JSPS KAKENHI (Grant Number JP222640019).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Arai, N., Tanaka, E., Ohnishi, S. et al. Intravenous administration of ex vivo expanded human umbilical cord blood-derived CD34⁺ cells in a preterm hypoxic-ischemic encephalopathy mouse model. Sci Rep (2026). https://doi.org/10.1038/s41598-026-48204-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-48204-z